

| Home |
| People |
| Research |
| Publications |
| News and Activity |
| Facilities |
| Visitors |
| Feedback |
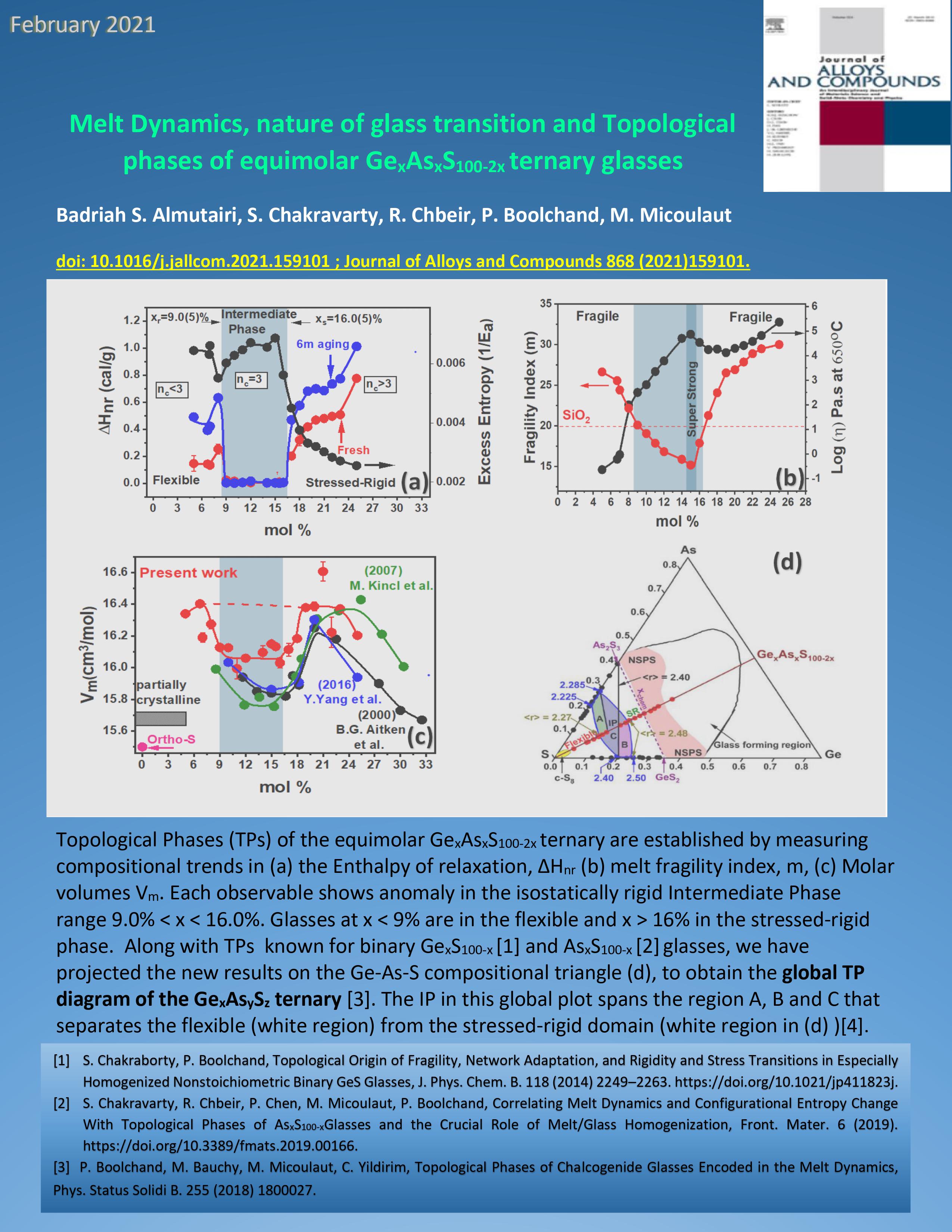
|
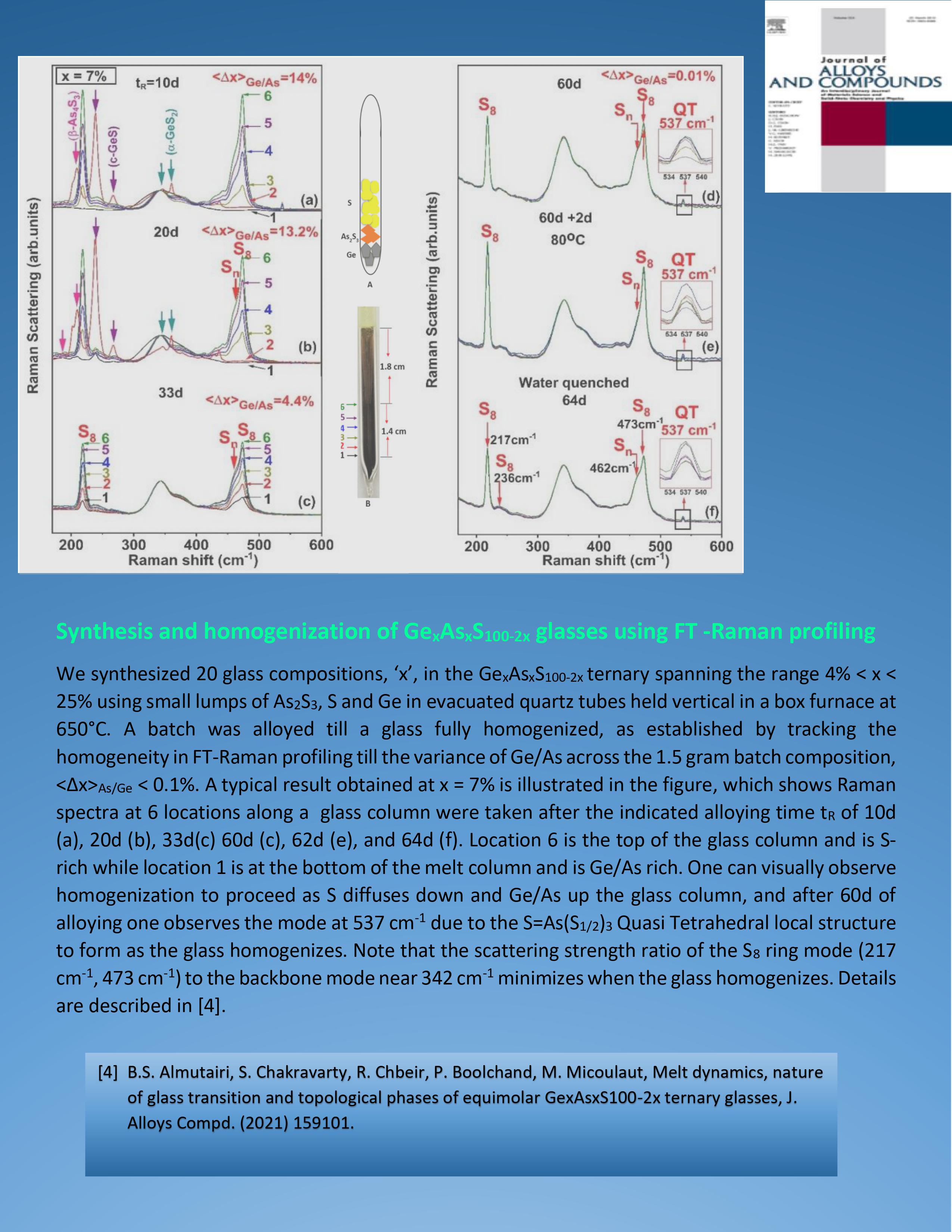
|
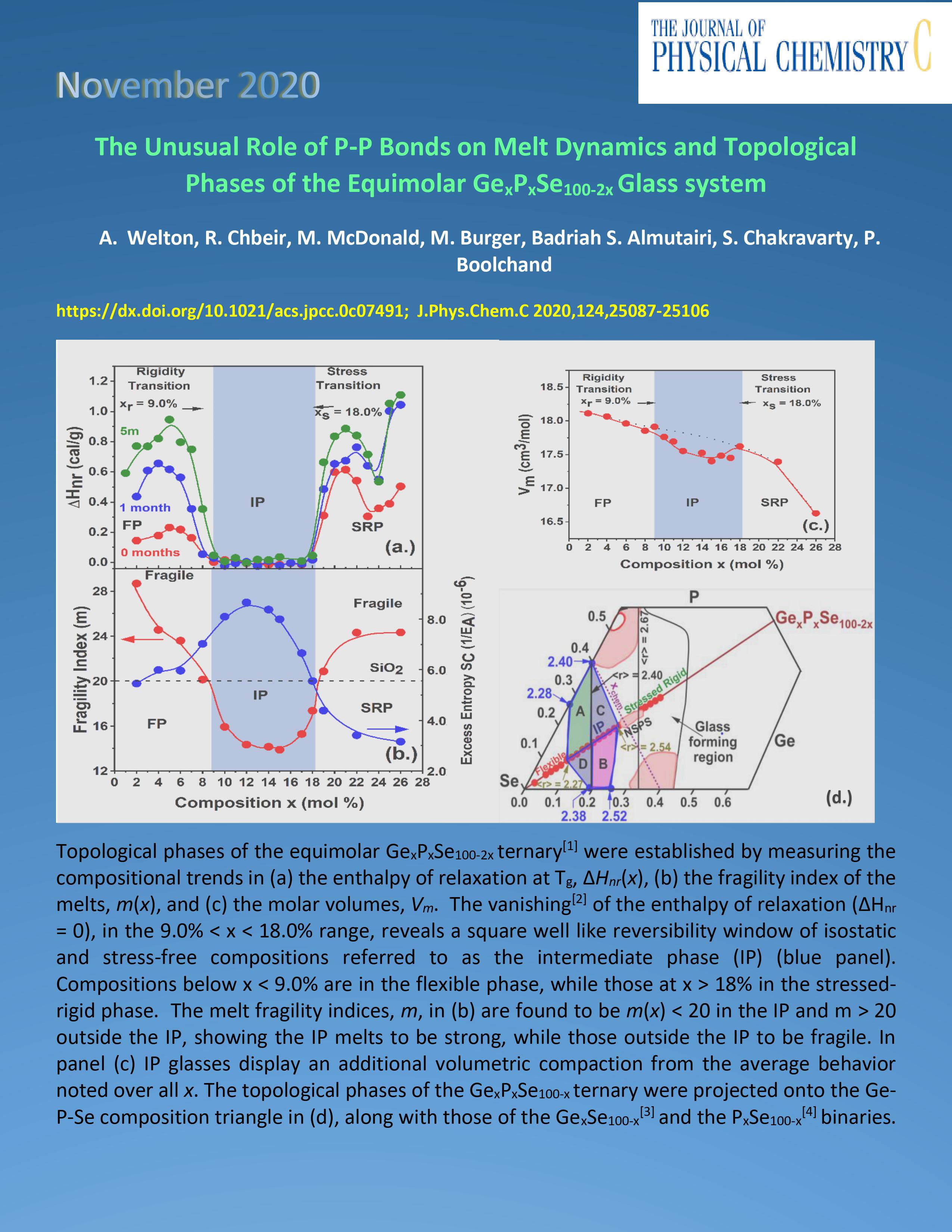
|

|
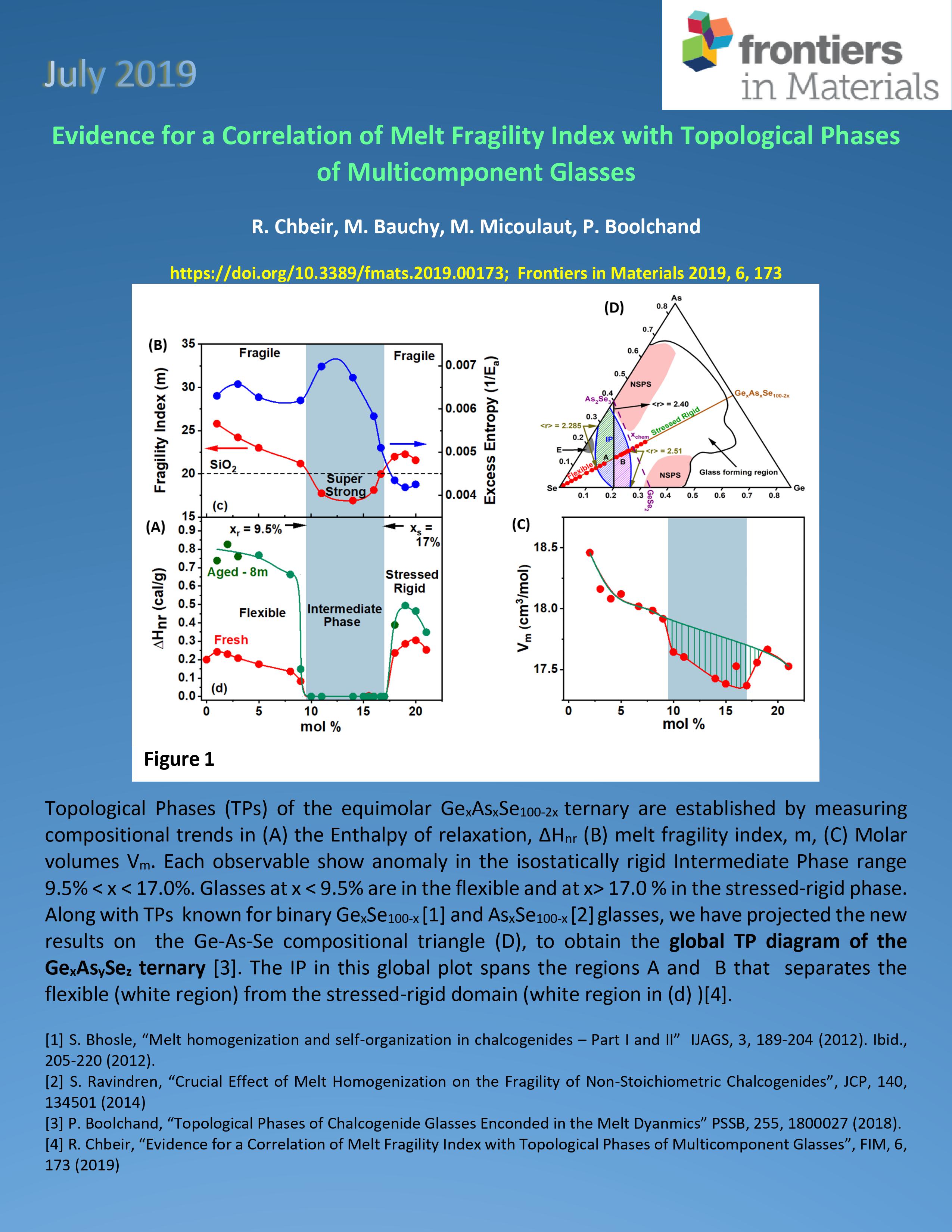
|
|
|
|
|
|
|
 |
|||||||||||||
 |
|
Research Interests | |||||||||||||
|
|||||||||||||||
|
|||||||||||||||